Apple AirPods Pro 2 Hearing Aid Features Gain FDA Approval: A Game Changer for Hearing Aids
Apple AirPods Pro 2 Hearing Aid Features Approval
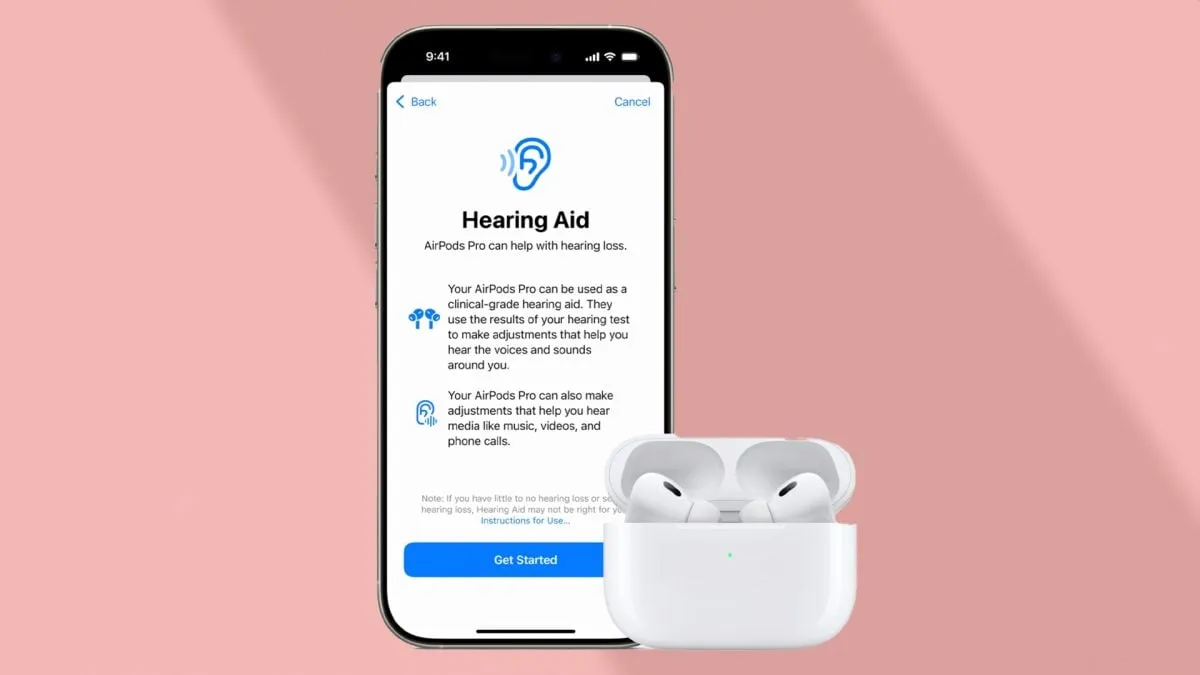
The US Food and Drug Administration (FDA) has officially approved clinical-grade hearing aid features for the Apple AirPods Pro 2, expected to launch via an upcoming software update. This transformative capability allows these popular audio devices to become practical solutions for individuals experiencing mild to moderate hearing loss.
Significance of the Approval
This groundbreaking FDA clearance signifies a pivotal moment as it provides an over-the-counter hearing support option. Previously, most hearing aids cost between $1,000 to $6,000, while the AirPods Pro 2 are priced at just $249.
- Michelle Tarver, MD, PhD, emphasized the need for affordable hearing solutions.
- The approval aims to assist millions of Americans affected by hearing loss.
Enhancements Through Software Update
Apple's software update will enhance the AirPods Pro 2's sound amplification capabilities, enabling users to adjust settings independently. A recent clinical study with 118 participants verified that these new features recaptured similar benefits as traditional hearing aids.
- Customizable sound amplifications.
- Real-time adjustments for environmental sounds.
This integration of hearing support into already popular consumer technology demonstrates Apple’s commitment to improving public health accessibility.
Future Implications and Rollout
The planned rollout for this new update is set for the end of 2024, with more than 100 countries expected to gain access. Users will leverage their iOS devices to tailor the experience based on their specific hearing needs.
This article was prepared using information from open sources in accordance with the principles of Ethical Policy. The editorial team is not responsible for absolute accuracy, as it relies on data from the sources referenced.